Current projects
GENETIC MARKERS
Large-scale genotyping of persons with Down syndrome in order to identify risk or protective factors of Alzheimer’s disease.
Despite the importance of Alzheimer’s disease (AD) in the population with Down syndrome (DS), there are currently no large-scale collaborative genomic studies of AD aimed at understanding the genes that determine the onset of AD.
The goal of the Horizon 21 consortium is to achieve a sample size with sufficient statistical power that is essential for the success of genetic association studies. The project aims at detecting the potentially causative genes involved in the pathogenesis of AD in people with DS.
In order to achieve this, the consortium shares clinical, epidemiological and molecular data.
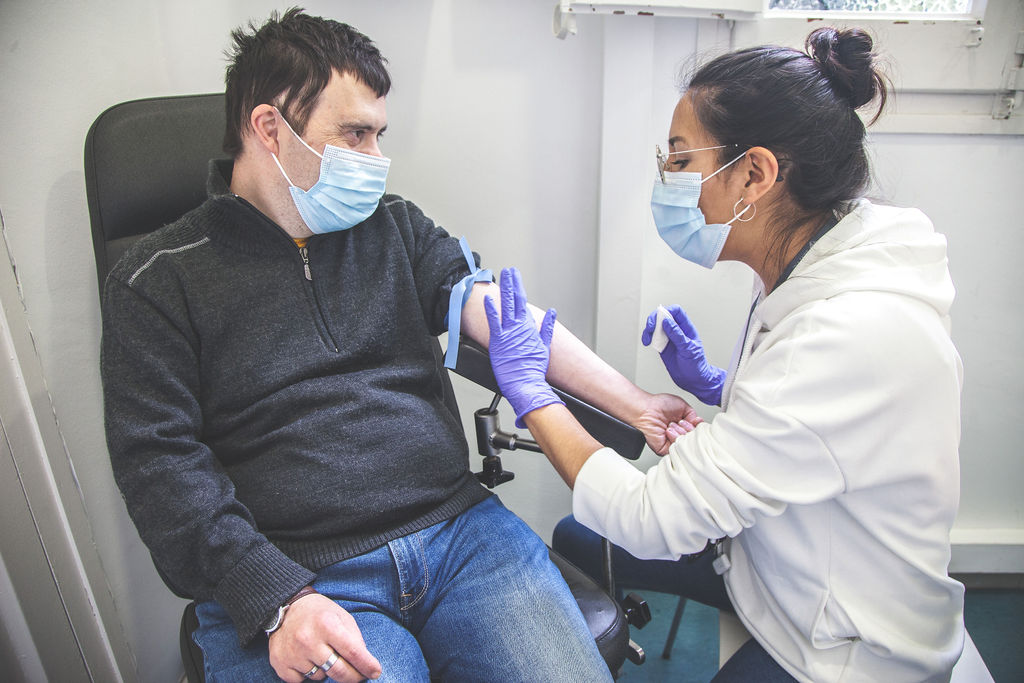
COGNITIVE EVALUATION TOOLS
Harmonisation of cognitive evaluation tools
Diagnosing dementia and assessing cognitive changes in adults with Down syndrome (DS) is difficult because of the presence of intellectual disability (ID) and the wide variability in functioning between individuals.
There is currently a critical lack of appropriate diagnostic measures and outcomes that can be used in clinical settings and randomized trials.
The challenge here is to better understand the pre-clinical and prodromal progression of AD in DS. Indeed, it is necessary to identify the characteristics of early AD-related changes in people with DS in order to design trials.
Cognitive testing is a key requirement for measuring clinical trial outcomes.
However, it remains to be seen whether these tools are sensitive enough to be used as outcome measures in clinical trials of treatment aimed at preventing disease progression.
These questions highlight the need to identify and describe in much greater detail the early cognitive and behavioural changes associated with AD, and to refine or develop more sophisticated neuropsychological tools to monitor cognitive decline in this population.
Longitudinal study of early markers of Alzheimer’s disease (AD) in adults with trisomy 21 (Horizon-21 cohort)
The main objective is to identify sensitive measures (at the clinical and biomarker level) of early cognitive decline that can be used as early markers of the neuropathology of AD, and to obtain longitudinal data on these markers in individuals with DS at risk for dementia.
The study involves the inclusion of 180 patients with Trisomy 21 who are at least 25 years of age in the 10 participating Horizon 21 consortium centers.

BIOLOGICAL MARKERS
e.g neurofilaments light analysis in plasma.
Several studies have shown that blood levels of neurofilaments (NfL) are increased in neurological disorders associated with neurodegeneration. This includes Alzheimer’s disease (AD), where increased NfL has been associated with mild cognitive impairment in addition to the cognitive, biochemical and brain imaging features of AD.
NfL has been identified as a possible blood biomarker of neurodegeneration associated with AD in Down syndrome (DS). It would allow the monitoring of disease progression while measuring cognitive and other outcomes.
The detection of abnormal levels of amyloid and tau in plasma is also a marker of interest.

SLEEP EVALUATION
Impact of Alzheimer’s disease on sleep of adults with Down syndrome.
In the general population, dementia is associated with more sleep disorders than those found in normal aging (nighttime awakenings, increased prevalence of behavioural disorders such as wandering, day/night confusion and nightmares or hallucinations, and daytime drowsiness with excessive napping and sleep-related breathing disorders).
Sleep disorders are very common in Alzheimer’s disease (AD) and have a negative impact on cognitive functions. They may be a risk factor in the progression of symptomatic AD.
Sleep problems are very common in people with AD. Obstructive Sleep Apnea (OSA) is the most common sleep disorder in the adult population with Down syndrome (DS) (78%). Sleep disorders have an impact on quality of life and affect physical abilities and mental health.
The hypothesis of this study is that adults with DS and AD will have a higher prevalence of sleep disorders (sleep disruption, circadian sleep disorders and OSA) than in DS without dementia.
